Overview of the Development of DprE1 Inhibitors for Combating the Menace of Tuberculosis | Journal of Medicinal Chemistry

Heterogeneity in non‐epitope loop sequence and outer membrane protein complexes alters antibody binding to the major porin protein PorB in serogroup B Neisseria meningitidis - Matthias - 2017 - Molecular Microbiology - Wiley Online Library

Organic and polymer chemistry of electrophilic tri‐ and tetrasubstituted ethylenes - Hall - 2004 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

Synthesis of B/Si Bidentate Lewis Acids, o-(Fluorosilyl)borylbenzenes and o-(Difluorosilyl)borylbenzenes, and Their Fluoride Ion Affinities | ACS Omega
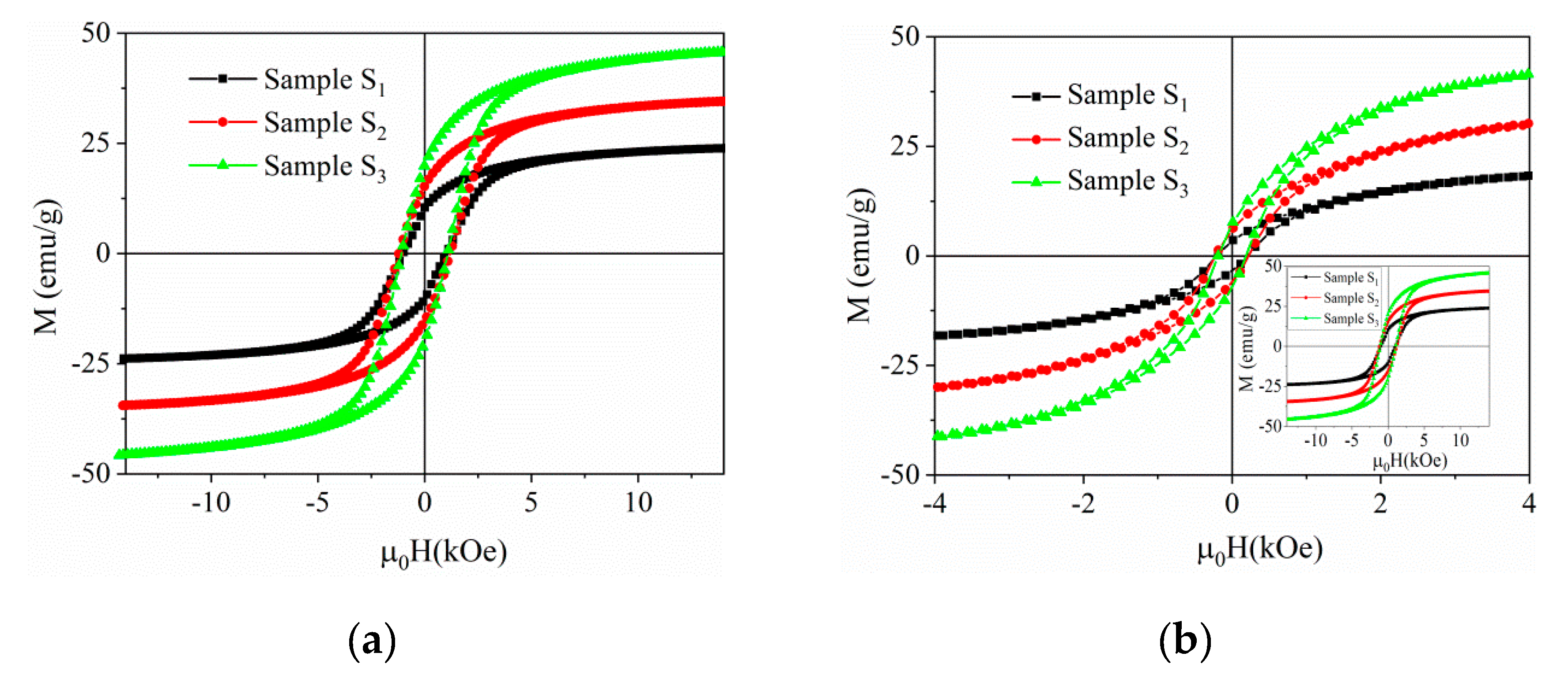
Nanomaterials | Free Full-Text | Lead-Free BNT–BT0.08/CoFe2O4 Core–Shell Nanostructures with Potential Multifunctional Applications

Theoretical Investigation of Substituent Effects on the Dihydroazulene/Vinylheptafulvene Photoswitch: Increasing the Energy Storage Capacity | The Journal of Physical Chemistry A

Organic and polymer chemistry of electrophilic tri‐ and tetrasubstituted ethylenes - Hall - 2004 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

Tylvalosin exhibits anti-inflammatory property and attenuates acute lung injury in different models possibly through suppression of NF-κB activation - ScienceDirect
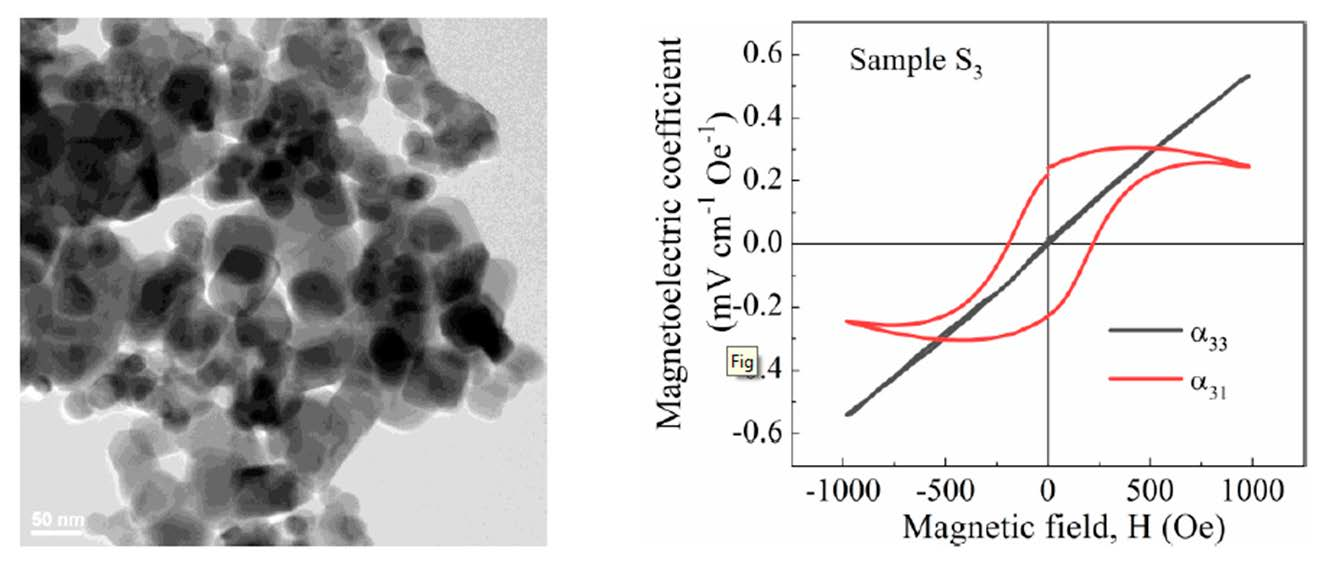
Nanomaterials | Free Full-Text | Lead-Free BNT–BT0.08/CoFe2O4 Core–Shell Nanostructures with Potential Multifunctional Applications

Synthesis of B/Si Bidentate Lewis Acids, o-(Fluorosilyl)borylbenzenes and o-(Difluorosilyl)borylbenzenes, and Their Fluoride Ion Affinities | ACS Omega
![Synthesis and Structure–Activity Relationships of Varied Ether Linker Analogues of the Antitubercular Drug (6S)-2-Nitro-6-{[4-(trifluoromethoxy)benzyl]oxy}-6,7-dihydro-5H-imidazo[2,1-b][1,3]oxazine (PA-824) | Journal of Medicinal Chemistry Synthesis and Structure–Activity Relationships of Varied Ether Linker Analogues of the Antitubercular Drug (6S)-2-Nitro-6-{[4-(trifluoromethoxy)benzyl]oxy}-6,7-dihydro-5H-imidazo[2,1-b][1,3]oxazine (PA-824) | Journal of Medicinal Chemistry](https://pubs.acs.org/cms/10.1021/jm200377r/asset/images/large/jm-2011-00377r_0014.jpeg)
Synthesis and Structure–Activity Relationships of Varied Ether Linker Analogues of the Antitubercular Drug (6S)-2-Nitro-6-{[4-(trifluoromethoxy)benzyl]oxy}-6,7-dihydro-5H-imidazo[2,1-b][1,3]oxazine (PA-824) | Journal of Medicinal Chemistry

PDF) Second-Order Nonlinear Optical Organic Materials: Recent Developments | Peter Gunter - Academia.edu

Heterogeneity in non‐epitope loop sequence and outer membrane protein complexes alters antibody binding to the major porin protein PorB in serogroup B Neisseria meningitidis - Matthias - 2017 - Molecular Microbiology - Wiley Online Library