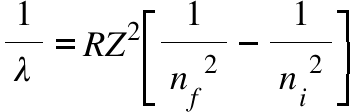Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (42 of 78) What is the Rydberg Constant? - YouTube
Positronium consist of an electron and a positron orbiting around their common centre of mass .calculate Rydberg constant for this system.

How to Calculate the Photon Frequency Absorbed or Emitted by an Electron in a Hydrogen Atom | Physics | Study.com

Physics - Ch 66.5 Quantum Mechanics: The Hydrogen Atom (42 of 78) What is the Rydberg Constant? - YouTube

Calculate the rydberg's constant,if the wavelength of first member of lyman series is 1215Å and hence find - Brainly.in

Using the Rydberg formula, calculate the wavelength of the first four spectral lines in the Lyma... - YouTube